Active Chest Tube Drainage System Clears the Way for Better Patient Care
By: Shana Leonard
Cardiothoracic surgeon Edward Boyle was a man on a mission when he partnered with Cleveland Clinic heart surgeon Marc Gillinov to establish Clear Catheter Systems Inc. (Bend, OR). After years of observing clogs and blood clots form in chest tubes and their resulting adverse effects on patients, Boyle was determined to develop a solution that overcame this dangerous design flaw. Responding to this unmet clinical need, Clear Catheter has introduced the PleuraFlow® Active Tube Clearance® System to prevent chest tube occlusion and improve patient safety.
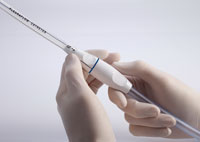
Following heart, lung, or trauma surgery, chest tubes are inserted into patients and coupled to a drainage system in order to remove excess fluid or air from the body. “A lot of what is drained is blood,” Boyle, CEO of Clear Catheter, explains. “If blood clots or coagulates in those tubes, which it tends to do, then they don’t drain very well. If that happens, it can contribute to complications and impair outcomes.” Complications resulting from clogged chest tubes can include a pneumothorax, pericardial tamponade, and excessive blood loss—the latter of which can occur if blood pools unnoticed in the chest cavity as a result of a clot formation in the portion of the tube positioned inside the body. Difficulty even arises when a clot is quickly identified in the chest tube, Boyle adds, because current methods of removal are undesirable for clinicians and may cause additional discomfort or risk for patients.
In an effort to avoid these unnecessary complications, Clear Catheter developed the PleuraFlow® system. The product consists of a standard chest tube that is inserted into the patient and then connected to a guide tube, over which is a shuttle guide set. The guide tube is then connected to a standard chest drainage system. Located within the guide tube, a guidewire featuring a distal loop moves backward and forward in the chest tube via a proprietary drive system to prevent clot formation. “We call this Active Tube Clearance®,” Boyle comments. “No other products have anything on the inside; they’re just a tube. With the PleuraFlow® system, we’re moving from passive drainage to active drainage.”
To develop the PleuraFlow® system, Clear Catheter Systems, armed with the initial concept for the clog-preventing device, sought the expertise of product development consultancy firm Carbon Design Group (Seattle) to help take the idea to the next level. Together, engineers and designers from both companies participated in brainstorm sessions to determine the optimal design of the system. “One of the most important design elements was that we had to figure out how to move the guidewire inside the tube from outside the tube,” Boyle recalls. “You can’t have an opening or a hole in the tube because that breaks the sterile barrier on the inside.”
A design epiphany came in the form of magnets, notes Robert Hubler, an industrial designer at Carbon. “There’s a magnet on the inside of the plastic tube that’s attached to the guidewire; on the outside, you have another magnet,” he explains. “The magnets will attract each other through the tube because the tube is naturally very slick on the inside and the magnet is a polished metal piece. So, it’s easy for the magnet to move on the inside of the tube with that attraction to the other magnet that’s moving back and forth without breaking the sterile barrier.” This back-and-forth movement of the guidewire in the tube serves to break up blockages, thus encouraging fluid flow and preventing coagulation.
Once the use of magnets was agreed upon, Carbon launched into the industrial design, mechanical engineering, and testing of the PleuraFlow® system based on feedback from Boyle and his surgeon colleagues. “Once Clear Catheter decided it wanted to go with the magnetic solution, we figured out a scheme for the use of the device pretty quickly,” Hubler says. “The basic architecture is that the shuttle gets released, moves back and forth, and then docks back in the home position.”
As part of the design and development process, Carbon took a close look at the forces required for the system. One challenge it faced was striking a balance so that the magnets provided enough force to drive the guidewire without interfering with nearby telemetry and other electronic systems. Achieving a balance in decoupling force also proved to be challenging. It needed to be strong enough to withstand the force of clot removal but forgiving enough to allow for separation of the wire in the shaft, if necessary, for safety reasons.
Upon finalizing the design and engineering with Carbon, Clear Catheter approached Xeridiem Medical Devices (Tucson, AZ), a company specializing in the design, development, and manufacture of complex, single-use medical devices. “We optimized the design for manufacturability,” says Mike Cusack, director of business development at Xeridiem. “We developed a manufacturing process and ensured that the device would work within the requirements of the ICU.”
Xeridiem also molded the silicone housings, and assembled, tested, packaged, and sterilized the device. The company helped with regulatory filings for the PleuraFlow® system as well. In essence, Cusack remarks, Xeridiem has served as the bricks and mortar of Clear Catheter Systems in that it manufactures the PleuraFlow® in addition to taking orders, providing technical support, answering phones, maintaining inventory, shipping for distribution, and invoicing.
Obtaining FDA approval in December, the PleuraFlow® system offers a simple solution to the common—and life-threatening—problem of chest tube clogging. And if that weren’t enough, it offers the additional benefit of enabling the use of smaller chest tubes. “Rather than using large chest tubes that look like a garden hose, our goal is to eventually be able to use a tube that is smaller and maybe only the size of a drinking straw,” Boyle says. “We think that it will not only help reduce complications, but it will also improve patient comfort.”