Blake Drains Can Cause RBC
For years, surgeons have been trying to improve the evacuation of post cardiac surgery blood to prevent retained blood complex (RBC). Various permutations of sucti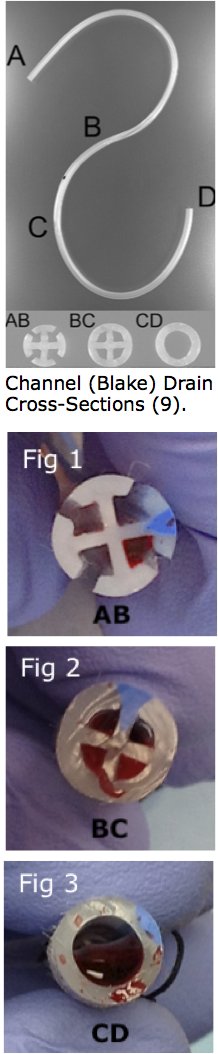 on drains (anticoagulant coatings, sump-style drains) have been tried, but the problem still remains
on drains (anticoagulant coatings, sump-style drains) have been tried, but the problem still remains that the single central lumen clogs with clot and impairs evacuation. (1-3) When this happens, acute complications can ensue, or sub-acute complications can result that slow recovery and contribute to readmissions.
Unsatisfied with conventional single lumen drainage tubes that frequently clotted off, cardiac surgeons started to explore different drain geometries with the hope of improving evacuation. Channel, or Blake drains, have multiple open channels inside the patient (AB), transition to a closed off section (BC), and finally to a complete tube (CD). The thought was that by providing multiple channels for the fluid to travel through, the tubes would be less susceptible to clogging.
In the open section of the drain (AB), evacuation is thought to work by “capillary” action. Suction in the closed section of the tube draws liquid into the tube, and the cohesive force between the liquid molecules perpetuates the evacuation. We’ve all observed this by watching water “climb” a paper towel, and this form of evacuation works great when evacuating less viscous fluid, like very thin serosanguinous fluid. However, blood in the post-surgical space undergoes a phase change from liquid to solid when coagulation is restored post operatively and the blood encounters the artificial materials of the chest tube. Now consider draining a jar of peanut butter with a paper towel: the oil will soak into the paper towel, but the cohesive attraction is not strong enough to move the solids, which are left behind. Similarly, when blood begins to coagulate, the channels are able to draw the liquid portion of the blood through the drain, but the solid portion is left behind (Fig 1). Channel (Blake) Drain Cross-Sections.This solid portion is a strong nidus for subsequent effusion production.
Compounding this problem is that at the transition point (BC), the channels form four small paths through which the blood must pass, each of which make up less than a quarter of the internal tube diameter. These paths can easily become clogged with thrombus (Fig. 2) and shut off the evacuation of blood from the chest. When blood finally reaches the tubular section of the drain (CD), it has the same propensity to clog the tube as it does in conventional tubular chest tubes (Fig 3). Though the exact incidence of channel drain clogging is unknown, life-threatening events have been reported because of channel drain clogging after thoracic surgery.(4) For these reasons, studies have shown that channel drains do not improve the evacuation of post surgical blood compared to conventional chest tubes.(5, 6)
So what does improve postoperative blood evacuation? PleuraFlow ACT has been shown not only to improve postoperative evacuation, but to reduce the volume of blood retained in the thorax, even in downsized chest tube systems.(7, 8) So after all the effort in tube innovation, the solution is really quite simple– proactively clear away the clots that form inside of the tube to improve evacuation, prevent retained blood, and clear the pathway to recovery.
- P. Kumar, D. McKee, M. Grant, J. Pepper. Phosphatidylcholine coated chest drains: are they better than conventional drains after open heart surgery? European Journal of Cardio-thoracic Surgery 11 (1997) 769–773.
- Beaudet, RL. New technique for drainage after cardiac surgery. J Thorac Cardiovasc Surg. 78 (1979) 119-22.
- Duncan C, Erickson R, Weigel R. Effect of chest tube management on drainage after cardiac surgery. Heart Lung. 1987;16(1):1-9.
- Clark G, Licker M, Bertin D, Spiliopoulos A (March 2007). “Small size new silastic drains: life-threatening hypovolemic shock after thoracic surgery associated with a non-functioning chest tube”. Eur J Cardiothorac Surg 31 (3): 566–8.
- Staffan Bjessmo, Susanne Hylander, Jenny Vedin, Dag Mohlkertb, Torbjo ?rn Ivert. Comparison of three different chest drainages after coronary artery bypass surgery — a randomised trial in 150 patients. European Journal of Cardio-thoracic Surgery 31 (2007) 372—375.
- Timothy L. Frankel; Peter C. Hill; Sotiris C. Stamou; Robert C. Lowery; Albert J. Pfister; Arvind Jain; and Paul J. Corso. Silastic Drains vs Conventional Chest Tubes After Coronary Artery Bypass. CHEST 124(2003) 108-113.
- Shiose, A.; Takaseya, T.; Fumoto, H.; Arakawa, Y.; Horai, T.; Boyle, E. M.; Gillinov, A. M.; Fukamachi, K. “Improved drainage with active chest tube clearance.” Interactive CardioVascular and Thoracic Surgery 10 (5): 685–688.
- Arakawa, Yoko; Shiose, Akira; Takaseya, Tohru; Fumoto, Hideyuki; Kim, Hyun-Il; Boyle, Edward M.; Gillinov, A. Marc; Fukamachi, Kiyotaka. “Superior Chest Drainage With an Active Tube Clearance System: Evaluation of a Downsized Chest Tube.” The Annals of Thoracic Surgery 91 (2): 580–583.
- Noriaki Sakakura, Takayuki Fukui, Shoichi Mori, Shunzo Hatooka, Kohei Yokoi and Tetsuya Mitsudomi. Fluid Drainage and Air Evacuation Characteristics of Blake and Conventional Drains Used After Pulmonary Resection?Ann Thorac Surg 2009;87:1539-1545